CHemistry
Chapter 1 - Matter & Change
Physical vs. Chemical Properties
Physical vs. Chemical Properties
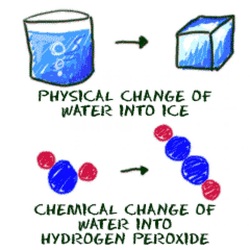
Physical Properties - a characteristic that can be observed or measured without changing the identity of the substance.
Each chemical has its own unique set of Physical Properties. It's kind of like a fingerprint for that chemical.
Examples: Boiling point, Melting point, Solubility
Each chemical has its own unique set of Physical Properties. It's kind of like a fingerprint for that chemical.
Examples: Boiling point, Melting point, Solubility
Separation Lab:
We can utilize the physical properties of different chemicals in order to separate them from other chemicals. Some of these separation strategies include:
Sublimation - the process of a solid evaporating directly into the gaseous phase (example: CO2, or dry ice)
Extraction - using the solubilities of different chemicals (example: sand & sugar, only sugar will dissolve in water)
Decanting - pouring away a liquid from a solid
Evaporation/Boiling - evaporate the chemical with the lower boiling point first, leaving the chemical with the lower BP
|
Guess what.... This video should look awfully familiar....... Watch it a few times and you'll be ready for your lab. |
|
Chemical Properties - a substance's ability to undergo changes that transform it into different substances.
The three ways to determine a CHEMICAL CHANGE occurred:
- Color Change
- Gas Produced
- Precipitate (solid) formed
Examples: oxidation (Fe rusting), combustion (burning)
Types of Matter
1. Pure Substances: Every sample has exactly the same properties and exactly the same composition.
Examples: Elements (Ca, Na, K, Cl), Compounds (NaCl, H2O, Ca(OH)2
2. Mixtures: Two or more chemicals mixed together.
A. Homogeneous - (also called solutions) uniform in composition
Examples: salt water (NaCl + H2O)
B. Heterogeneous - not uniform in composition. Heavier particles may sink to bottom.
Examples: muddy water
The Periodic table
|
|
Here are some videos discussing some of the trends and different elements found on the periodic table.
Some of the Key Terms needed from the Periodic Table:
|
|
Dmitri Mendeleev
This was the guy who put together the first periodic table, and this allowed him to PREDICT NEW ELEMENTS !!! |
|
|
|
And.....
I really like this guy, so here is another video by this guy...... about the periodic table. |
|
Also, you will notice that there are a lot of songs/raps all about the periodic table.
Here is a new one I just found. Not saying it's the best out there. Just a newer one. YouTube "periodic table song" or "rap" and you'll find a whole bunch of funny, nerdy, weird, and just creepy periodic table stuff. |
|
