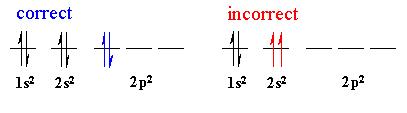Light
It travels through space as WAVES.
It is VISIBLE. It is INVISIBLE.
And it's Radiation.
Sounds fun, huh? But the light we see and rely on to "see" is all ELECTROMAGNETIC RADIATION.
Electromagnetic Radiation: Energy that travels through empty space.
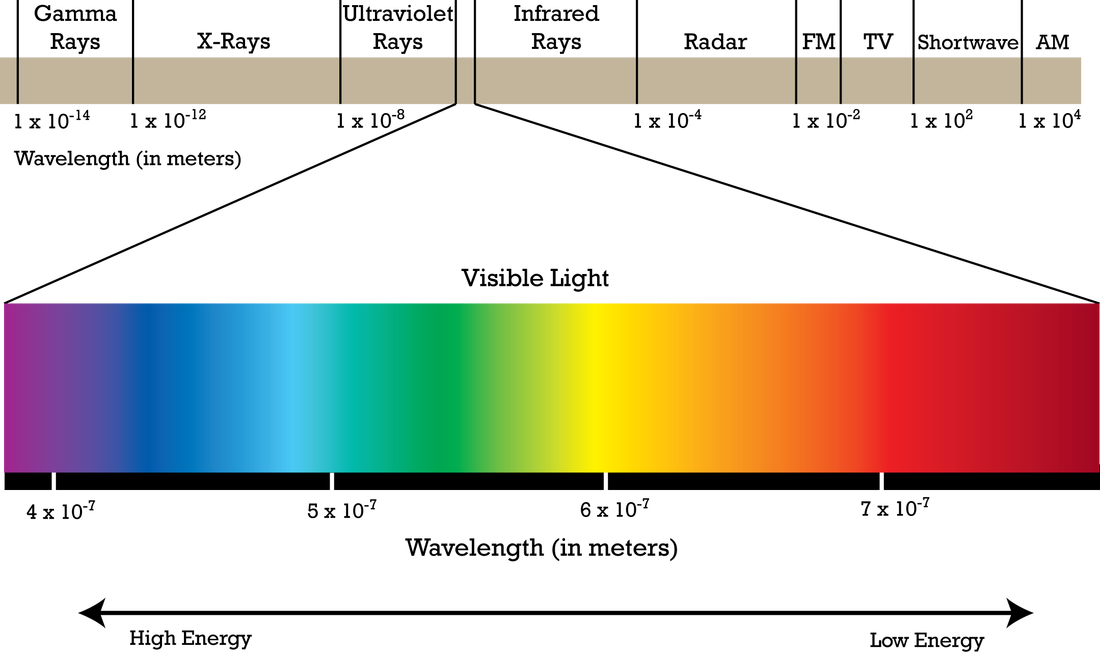
Below is a graphic showing the different types of electromagnetic radiation, or light.
It travels through space as WAVES.
It is VISIBLE. It is INVISIBLE.
And it's Radiation.
Sounds fun, huh? But the light we see and rely on to "see" is all ELECTROMAGNETIC RADIATION.
Electromagnetic Radiation: Energy that travels through empty space.
Below is a graphic showing the different types of electromagnetic radiation, or light.
The photoelectric effect
This was one of the experiments that scientists conducted back in the early 1900's that could not be explained by the wave theory of light.
This is where light (electromagnetic radiation) is shined on a metal. The light causes electrons to be emitted from the metal.
The frequency of the light (color, or energy of the light) determines whether or not electrons would or could be emitted from the metal's surface.
Below is a video showing the PHOTOELECTRIC EFFECT
This is where light (electromagnetic radiation) is shined on a metal. The light causes electrons to be emitted from the metal.
The frequency of the light (color, or energy of the light) determines whether or not electrons would or could be emitted from the metal's surface.
Below is a video showing the PHOTOELECTRIC EFFECT

Max Planck (1858-1947)
Quantum of Energy : minimum quantity of energy that can be lost or gained by an atom.
Planck's relationship between energy & frequency --
E = h * v
E = energy v = frequency h = Planck's constant (6.626x10^-34 J*s)
- Proposed that a hot object does not emit electromagnetic radiation/energy continuously
- Suggested that energy is emitted in small, specific packets called quanta.
Quantum of Energy : minimum quantity of energy that can be lost or gained by an atom.
Planck's relationship between energy & frequency --
E = h * v
E = energy v = frequency h = Planck's constant (6.626x10^-34 J*s)

Albert Einstein (1879-1955)
Among his many accomplishments, discoveries, and theories, he expanded upon Planck's theory of quanta by suggesting that electromagnetic radiation has a dual wave-particle nature. That is.....LIGHT exhibits the properties of both a particle AND a wave at the same time.
The ENERGY of a photon is directly proportional to its FREQUENCY:
Ephoton = h * v
Among his many accomplishments, discoveries, and theories, he expanded upon Planck's theory of quanta by suggesting that electromagnetic radiation has a dual wave-particle nature. That is.....LIGHT exhibits the properties of both a particle AND a wave at the same time.
The ENERGY of a photon is directly proportional to its FREQUENCY:
Ephoton = h * v

Niels Bohr (1885-1962)
Developed the "energy model" of the atom, showing how electrons fill separate energy levels. As electrons absorb energy, they jump from the ground state (lowest possible) to an excited state (something above the ground state).
As the electron falls from the excited state to the ground state, it emits, or releases, the energy it had absorbed. The energy emitted can be observed as light.
Developed the "energy model" of the atom, showing how electrons fill separate energy levels. As electrons absorb energy, they jump from the ground state (lowest possible) to an excited state (something above the ground state).
As the electron falls from the excited state to the ground state, it emits, or releases, the energy it had absorbed. The energy emitted can be observed as light.
The quantum model
Light can be both a PARTICLE and a WAVE.

Louis DeBroglie (1892-1987)
Suggested that electrons (like light) exist as both particles & waves. That changed our understanding of matter. This idea won him the 1929 Noble Prize in Physics.
Suggested that electrons (like light) exist as both particles & waves. That changed our understanding of matter. This idea won him the 1929 Noble Prize in Physics.
|
Werner Heisenberg (1901-1976)
Most well known for his Uncertainty Principle from 1927, Heisenberg was one of the groundbreakers in quantum mechanics. He won the 1932 Nobel Prize in Physics 'for the creation of quantum mechanics' and his neutron-proton model of the nucleus. **Uncertainty Principle** ~ It is impossible to determine simultaneously both the position AND velocity of an electron. |
|
Erwin Schrödinger (1887-1961)
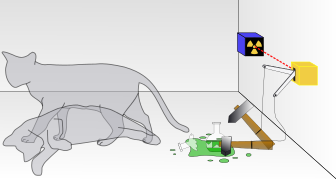
A key figure in quantum mechanics, Schrodinger is most well known for his cat. Schrödinger's cat: a cat, a flask of poison, and a radioactive source are placed in a sealed box. If an internal monitor detects radioactivity (i.e. a single atom decaying), the flask is shattered, releasing the poison that kills the cat. The Copenhagen interpretation of quantum mechanics implies that after a while, the cat is simultaneously alive and dead. Yet, when one looks in the box, one sees the cat either alive or dead, not both alive and dead. This poses the question of when exactly quantum superposition ends and reality collapses into one possibility or the other. |
orbitals
3D region around the nucleus of an atom that indicates the probable location of electrons.
Quantum Numbers
Quantum Numbers
- Principle quantum number (n) - indicates the energy level occupied by electron (ex: 1, 2, 3) - also called shells
- Angular Momentum Quantum number (l) - indicates the shape of the orbital
- Magnetic Quantum number (m) - indicates orientation around nucleus.
- Spin Quantum number - electrons spin like earth on axis, creating a magnetic field. The spin # was created to account for that magnetic field. (+1/2 or -1/2)
Electron configuration

The arrangement of electrons in an atom (in ground state).
3 Governing Rules
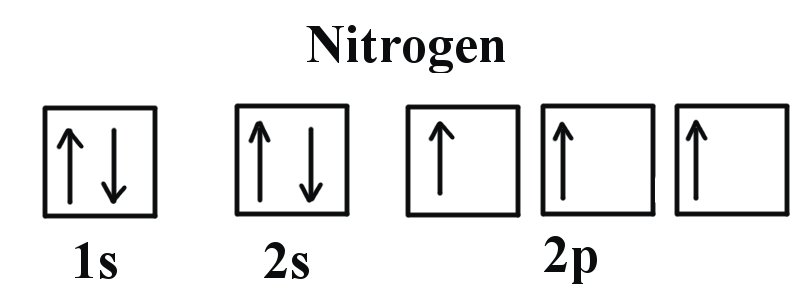
1. Aufbau principle - electrons occupy the lowest energy orbital that can receive it.
(starts at E=1 (n=1) and continues up)
3 Governing Rules
1. Aufbau principle - electrons occupy the lowest energy orbital that can receive it.
(starts at E=1 (n=1) and continues up)